Unveiling the Significance of Trend Reporting Under Article 88
Article 88 of the EU MDR introduces a critical mechanism for monitoring and addressing trends in medical device safety data — and reshapes how manufacturers report.
A Paradigm Shift in Post-Market Surveillance
With the implementation of the European Medical Device Regulation (EU MDR), the regulatory landscape underwent a paradigm shift, placing unprecedented emphasis on proactive post-market surveillance (PMS). Among its most significant provisions is Article 88, which specifically mandates trend reporting.
This represents a fundamental move away from merely reacting to individual severe incidents, towards identifying subtle, emerging patterns that could indicate systemic issues before they result in patient harm.
What Does Article 88 Actually Require?
Article 88 requires manufacturers to report any statistically significant increase in the frequency or severity of non-serious incidents or expected undesirable side-effects. This requirement is complex because it demands:
- Continuous, rigorous statistical analysis of post-market data.
- Establishing precise baseline rates for all device models.
- Setting actionable thresholds for deviation.
Manufacturers can no longer simply log complaints; they must actively monitor these baselines and have systems in place to detect deviations in real-time.
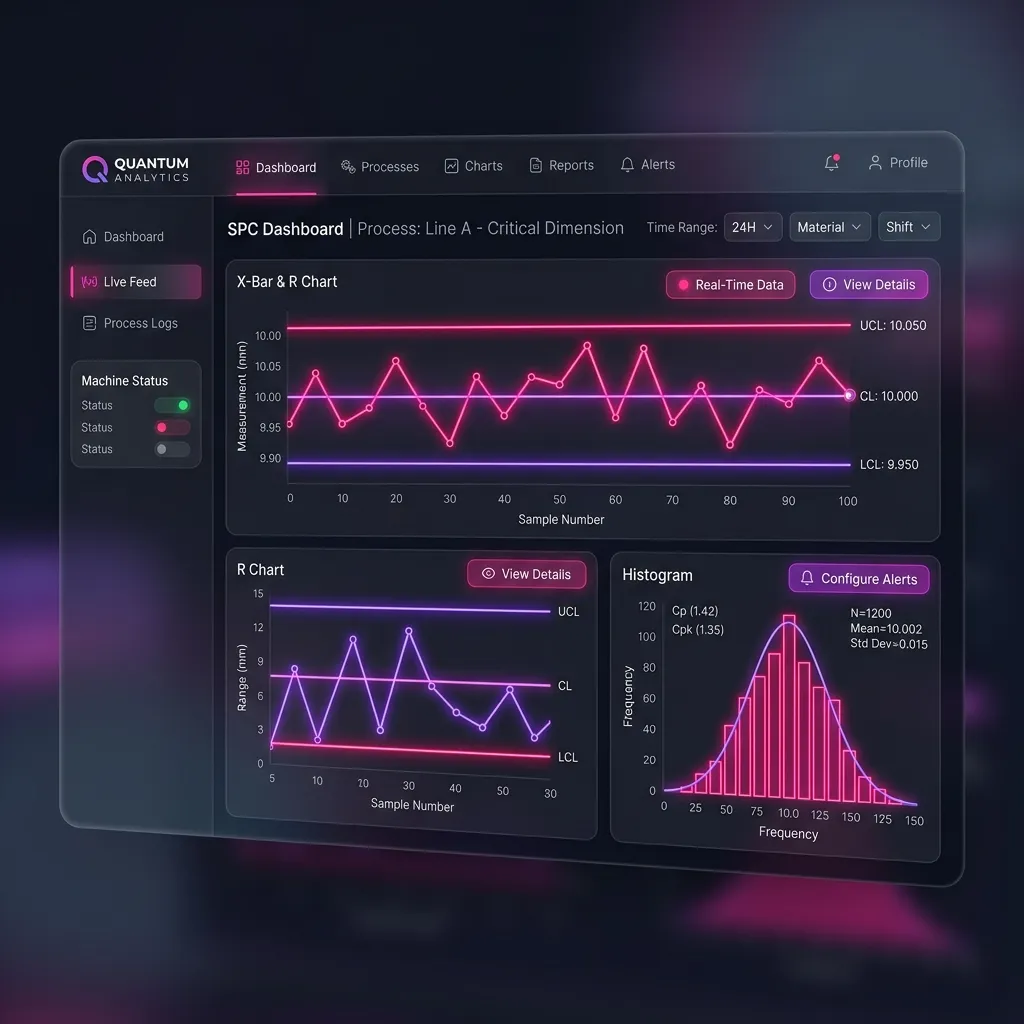
The Analytical Challenge
The challenge lies in the sheer volume and variability of the data. Determining what constitutes a "statistically significant" trend requires robust analytical capabilities that traditional spreadsheets simply cannot handle.
"In regulated contexts, 'mostly correct' manual analysis has an annoying habit of becoming completely unreliable during an audit."
This is where modern data management platforms become indispensable. By utilizing advanced analytics and automated threshold monitoring, companies can ensure they are not only compliant with Article 88 but are genuinely improving patient safety by catching potential risks before they escalate into major incidents.
Ready to automate your trend reporting? Let's connect to see how Data Reality handles complex statistical modeling for MedTech.
Ready to streamline your compliance?
Discover how Data Reality's AI-driven platform can transform your regulatory workflows today.
Get a Demo