Data Reality's Automated AI-Based PSUR
Reimagining the Periodic Safety Update Report. The PSUR is a cornerstone of post-market surveillance — and a notorious bottleneck for regulatory teams. Here's how AI is collapsing months of work into hours.

The Regulatory Bottleneck No One Wants to Talk About
The Periodic Safety Update Report (PSUR) is arguably one of the most critical elements of post-market surveillance for medical device manufacturers under the EU MDR (Article 86). It provides a comprehensive evaluation of the risk-benefit balance of a device throughout its lifecycle.
However, the traditional process of compiling a PSUR is notoriously labor-intensive. It requires regulatory teams to sift through mountains of clinical data, complaints, literature, and post-market clinical follow-up (PMCF) data.
Teams are asking the wrong question
"Can't we just hire more medical writers?"
That conversation is already old news in most organizations. Many teams are already tapped out on resources, facing burnout, and struggling to meet strict Notified Body deadlines.
The better question is:
"What kind of automation is appropriate for this highly regulated task?"
Enter Data Reality's Automated AI-Based PSUR Generation
By leveraging advanced natural language processing (NLP) and machine learning algorithms trained specifically on MedTech terminology, our platform automates the ingestion and analysis of disparate data.
- Data Ingestion: Automatically connects to your eQMS, ERP, and CRM to pull complaint data.
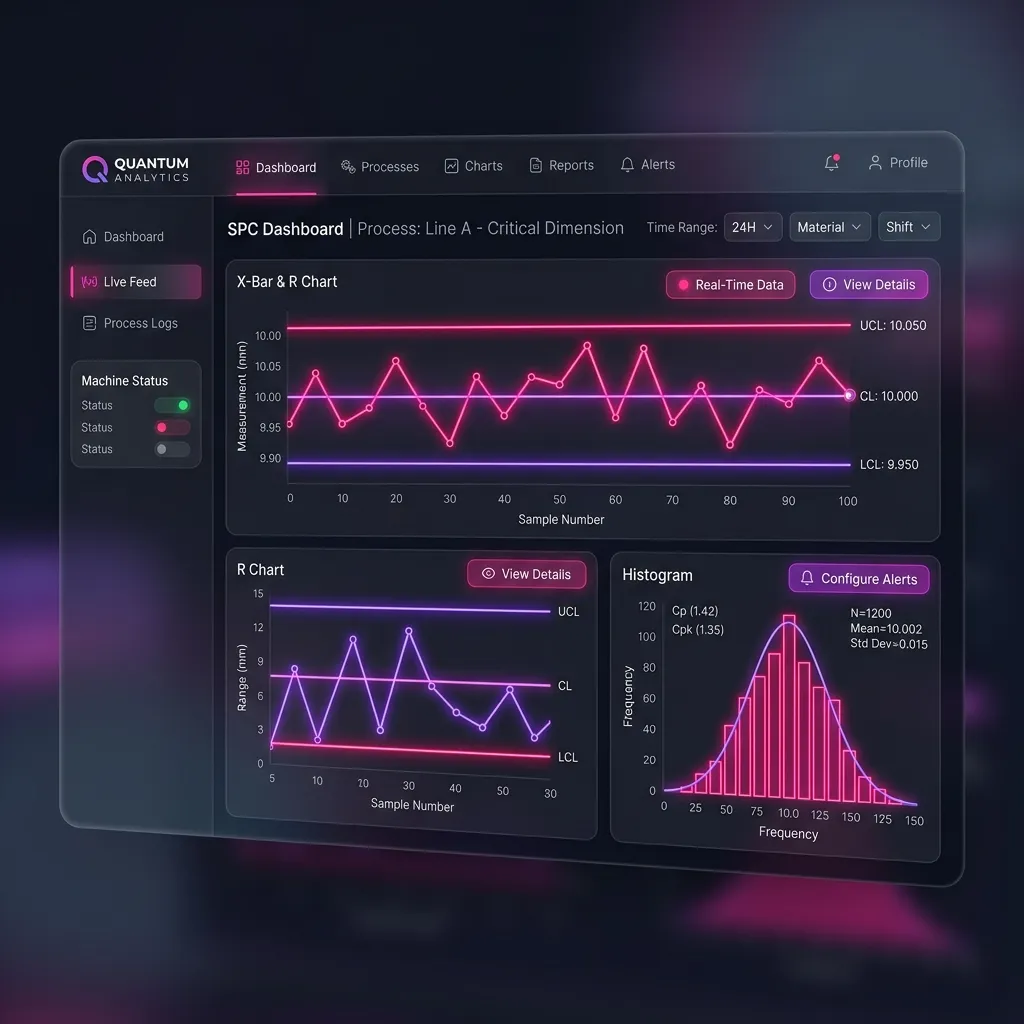
- Signal Detection: Intelligently synthesizes inputs, identifying safety signals and calculating adverse event rates.
- Literature Review: Scrapes PubMed and other databases to summarize the state of the art.
This transformation collapses what used to take weeks or months of manual effort into mere hours. The result is a highly accurate, compliant draft report ready for expert human review.
Is 80% Accuracy Good Enough?
In many industries, absolutely! If a generic AI gets you most of the way there and a human cleans up the rest, that's a fine trade.
But in MedTech? Outputs become part of regulatory documentation, where small errors have massive compliance consequences. That is why Data Reality relies on Specialized AI built exclusively for medical devices. We aim for near-100% reliability, providing full traceability back to the source data.
If your team is navigating these regulatory hurdles right now, feel free to reach out anytime! We would love to discuss how our platform can fit into your specific workflow.
Ready to streamline your compliance?
Discover how Data Reality's AI-driven platform can transform your regulatory workflows today.
Get a Demo